Red and Far Infrared Fiber Lasers
-
 MPBC has a complete line of Red and Deep Red visible fiber lasers, the earliest models developed for the cutting-edge Microscopy platforms built in the laboratories of Dr. Stefan Hell, and flow Cytometry platforms built in the labs of Dr. William Telford of the Core Flow Cytometry Facility at N.I.H. where beam quality, reliability, and stability continue to be of the utmost importance.
MPBC has a complete line of Red and Deep Red visible fiber lasers, the earliest models developed for the cutting-edge Microscopy platforms built in the laboratories of Dr. Stefan Hell, and flow Cytometry platforms built in the labs of Dr. William Telford of the Core Flow Cytometry Facility at N.I.H. where beam quality, reliability, and stability continue to be of the utmost importance. -
Red and Deep Red Visible Fiber Lasers are air cooled, available in either an OEM compact package, and / or a 2RU rack mountable package with integrated power supply and are shipped with a user friendly graphical user interface for ease of set up and operation.
-
-
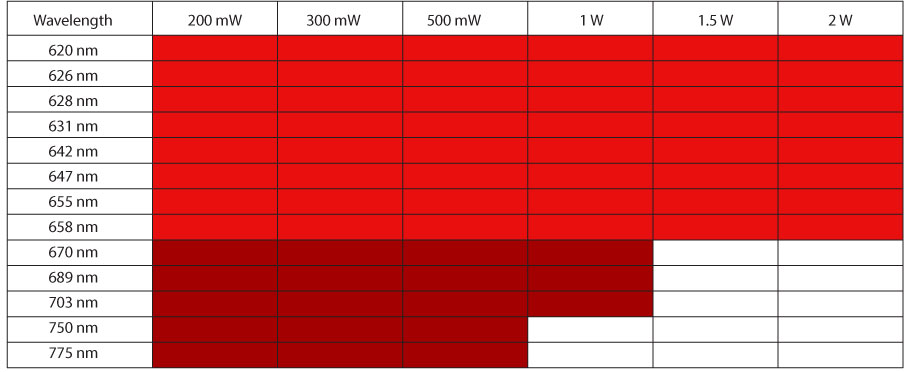
MPBC Fiber Laser Technology is so flexible we are able to offer our customers virtually any wavelength in the red spectrum between 620 nm (1240 nm) to 790 nm (1580 nm). To do this we employ our own fiber Bragg gratings to lock the laser wavelength and can produce in a very short turn-around time specific wavelengths addressing your requirement. If you have an application for a laser that is not covered in our current product portfolio, please reach out to us as we work closely with researchers to develop lasers to meet next generation requirements.
-
Features
- Narrow Linewidth
- Active Power Stabilization to ensure long term power stability of < 2%
- Excellent wavelength stability to ensure spectral purity (± 0.02 nm)
- Graphical User Interface for easy command and control
- Compact laser head
- Tunable output power (from 20% to 100% of nominal) to adapt to application-specific requirements
- High reliability
- Maintenance-free
Applications
- Flow Cytometry
- Fluorescence Microscopy
- Structured Illumination Microscopy
- Super Resolution Microscopy
- 3rd Generation DNA Sequencing
- DNA Painting
- Biomedical Research
- Entertainment
- Military, and Scientific Research
Specifications
- Beam: Collimated
- Transverse Mode: TEM00
- Polarization: Linear
- Orientation of Polarization: Vertical to the base
| Red Visible Fiber Lasers | Minimum | Nominal | Maximum | |
|---|---|---|---|---|
| Emission Wavelengths | 628 to 775* | mW | ||
| Output Power Range | 200 to 2000* | mW | ||
| Power Tunability Range` | 20 | 100 | % | |
| Ellipticity | 7 | 20 | % | |
| Beam Quality M2 | 1.1 | 1.15 | ||
| Emission Linewidth (FWHM) | 0.1 | nm | ||
| Long Term Wavelength Stability | Nominal - 0.02 | Nominal + 0.02 | nm | |
| Degree of Polarization | 98-99* | % | ||
| RMS noise at nominal power integrated over 5 Hz to 100 kHz |
2 | % | ||
| Long Term Outpower Drift (8 hours) peak to peak | 2 | % | ||
| * model-dependent. Contact MPBC for more detailed specifications. | ||||
Fiber Coupling Option
For clients who require a fiber delivered beam, MPB offers optional fiber coupling with a maximum output power from the fiber for our Red lasers of 1 Watt.
Single mode PM Patch cord output options include
- Bare fiber
- FC/PC connector (flat 0°)
- FC/APC connector (flat, angled 8°).
Download our “Visible Fiber Lasers for Bioscience” brochure for more information on this product line and what MPBC can do for you.
For more information on making a laser selection, see “Why M2 is Important When Selecting Your Laser.”
See how some of our customers are using our Red Visible Fiber Lasers
Microscopy
- Alan M. Szalai, Lucía F. Lopez, Miguel Ángel Morales-Vásquez, Fernando D. Stefani and Pedro F. Aramendí, "Analysis of sparse molecular distributions in fibrous arrangements based on the distance to the first neighbor in single molecule localization microscopy." Nanoscale, Issue 17, 2020.
- Elbaz-Alon, Y., Guo, Y., Segev, N. et al. "PDZD8 interacts with Protrudin and Rab7 at ER-late endosome membrane contact sites associated with mitochondria." Nature Communications 11, 3645 (2020). https://doi.org/10.1038/s41467-020-17451-7.
- Morozumi, A., Kamiya, M., Urano, Y., "Single-Molecule Localization Microscopy Propelled by Small Organic Fluorophores with Blinking Properties." Single Molecule Microscopy in Neurobiology (pp.203-227).
- Felix Wäldchen, F., Schlegel, J., Götz, R., Luciano, M., Schnermann, M., Doose, S. & Sauer, M. "Whole-cell imaging of plasma membrane receptors by 3D lattice light-sheet dSTORM." Nature Communicationsvolume 11, Article number: 887 (2020).
- Zhang, Y., Schroeder, L.K., Lessard, M.D. et al. "Nanoscale subcellular architecture revealed by multicolor three-dimensional salvaged fluorescence imaging." Nature Methods 17, 225–231 (2020). https://doi.org/10.1038/s41592-019-0676-4.
- Sapoznik E., Chang B-J., Huh J., Ju RJ., Azarova EV., Pohlkamp T., Welf ES., Broadbent D, Carisey A. "A versatile oblique plane microscope for large-scale and high-resolution imaging of subcellular dynamics." eLifeSciences.org https://elifesciences.org/articles/57681 November, 2020.
- Sha An, Karl Ferdinand Ziegler, Peiyi Zhang, Yu Wang, Tim Kwok, Fan Xu, Cheng Bi, Sandro Matosevic, Peng Yin, Tongcang Li, and Fang Huang, "Axial plane single-molecule super-resolution microscopy of whole cells," Biomed. Opt. Express 11, 461-479 (2020)
- Liu, S., Huang, F. Enhanced 4Pi single-molecule localization microscopy with coherent pupil based localization. Commun Biol 3, 220 (2020). https://doi.org/10.1038/s42003-020-0908-2
-
Etai Sapoznik, Bo-Jui Chang, Robert J. Ju, Erik S. Welf, David Broadbent, Alexandre F. Carisey, Samantha J. Stehbens, Kyung-min Lee, Arnaldo Marín, Ariella B. Hanker, Jens C. Schmidt, Carlos L. Arteaga, Bin Yang, Rory Kruithoff, Doug P. Shepherd, Alfred Millett-Sikking, Andrew G. York, Kevin M. Dean, Reto Paul Fiolka, "A Single-Objective Light-Sheet Microscope with 200 nm-Scale Resolution," bioRxiv 2020.04.07.030569; doi: https://doi.org/10.1101/2020.04.07.030569,
Now published in eLife doi: 10.7554/eLife.57681
Lattice Light Sheet
- Bin Cao, Guanshi Wang, Jieru Li, Alexandros Pertsinidis, "3D Interferometric Lattice Light-Sheet Imaging," doi: https://doi.org/10.1101/2020.08.27.266999
Genomics
- Liu M., Lu Y., Yang B., Chen Y., Radda J.S.D., Hu M., Katz S.G. & Wang S. "Multiplexed imaging of nucleome architectures in single cells of mammalian tissue." Nature Communications 11, 2907 (2020).
- Goh, JJL., Chou, N., Seow, WY., Ha, N., Cheng, CPP, Yun-Ching Chang, Y-N, Zhao, ZW & Kok Hao Chen , KH. "Highly specific multiplexed RNA imaging in tissues with split-FISH." Nature Methods volume 17, pages 689–693(2020).
Optogenomics
- McElmurry K., Stone JE., Ma D., Lamoureux P., Zhang Y., Steidemann M., Fix L., Huang F., Miller K.E., Suter DM. "Dynein-mediated microtubule translocation powering neurite outgrowth in chick and Aplysia neurons requires microtubule assembly." Journal of Cell Science 2020 133: jcs232983 doi: 10.1242/jcs.232983 Published 24 April 2020

